While visiting Riken in Wako, Japan when I was on exchange in Tokyo, I encountered an interesting question. The professor showing me around mentioned how one of the ion beams they were using in one of the accelerators were fully ionized large atoms of Rhenium . These atoms were accelerated and used to create atoms of a new element, Nihonium.
With a background in Chemistry this struck me as very surprising. In regular Chemistry the highest charged ions one comes across is something like , and even then the charge is never fully bare.
Hence, a charge of +75 was very surprising to me. The process to achieve this is impressive, and Riken routinely generates heavy ions like with multiple stripper stages. However, I had a different question. What is the most highest ionization state physically possible? And if there is a limit, is it only one of technology, or fundamental?
There's actually a pretty nifty answer I found, although I'm not the first.
Starting with the Rydberg equation for the energy states of a bare atom with charge , we can calculate the energy of the lowest orbital, the electron affinity of this atom. This is a rough estimation, since for about relativistic effects start to become important. A simple 1st order relativistic correction (fine-structure correction) helps a bit, but the full relativistic Dirac equation is needed to get an accurate result. Here are the results from a Matlab script plotting this energy for increasing :
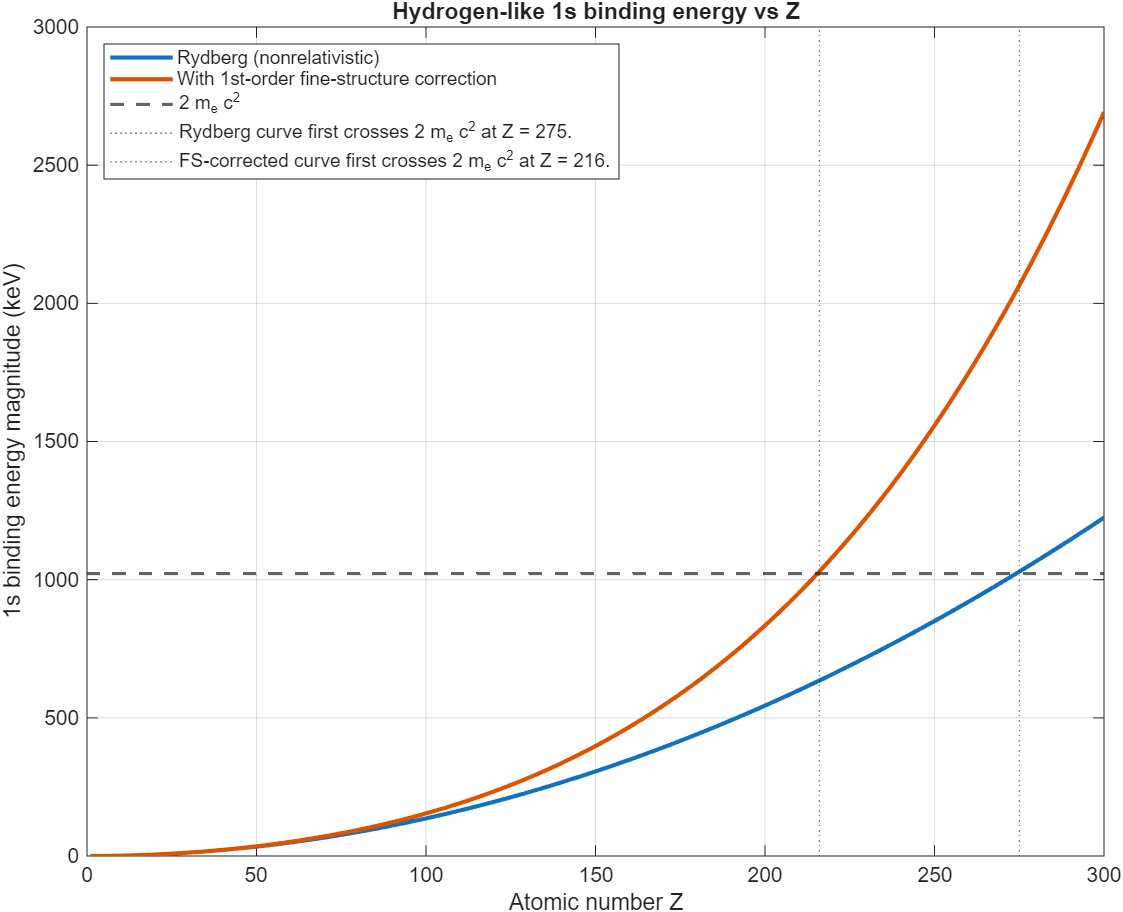
For both, the electron affinity reaches twice the electron mass. For Rydberg, this happens at , with relativistic correction this number drops to . This line is interesting, since at this point forming a electron-positron particle pair and then binding the electron while ejecting the positron becomes energetically favorable. This means that if such an ion were to exist, it would immediately decay by binding an electron and emit a positron. Cool!
This can also be understood through the Schwinger limit, i.e. there being a maximum possible electric field. Above that field strength, electron-positron pairs will form, remove energy from the field and screen it. Fields stronger than this rapidly decay through the same mechanism as in our ion.
As more relativistic corrections are added, the maximum number drops further, see below. In addition to the relativistic effects on the electron, the Coulomb potential near a highly charged ion itself is also modified. Similar to the fine-structure correction, the first order correction here takes the form of an Uehling potential.
Using the Dirac equation, a similar issue appears for atoms of . There are claims that heavy atoms beyond this atomic number are not possible due to this. Here screenshots from the Wikipedia article on the Extended periodic table. Worth a read if you find this question interesting!
Wherever exactly this limit might be, that it exists is an interesting little fact about our universe.